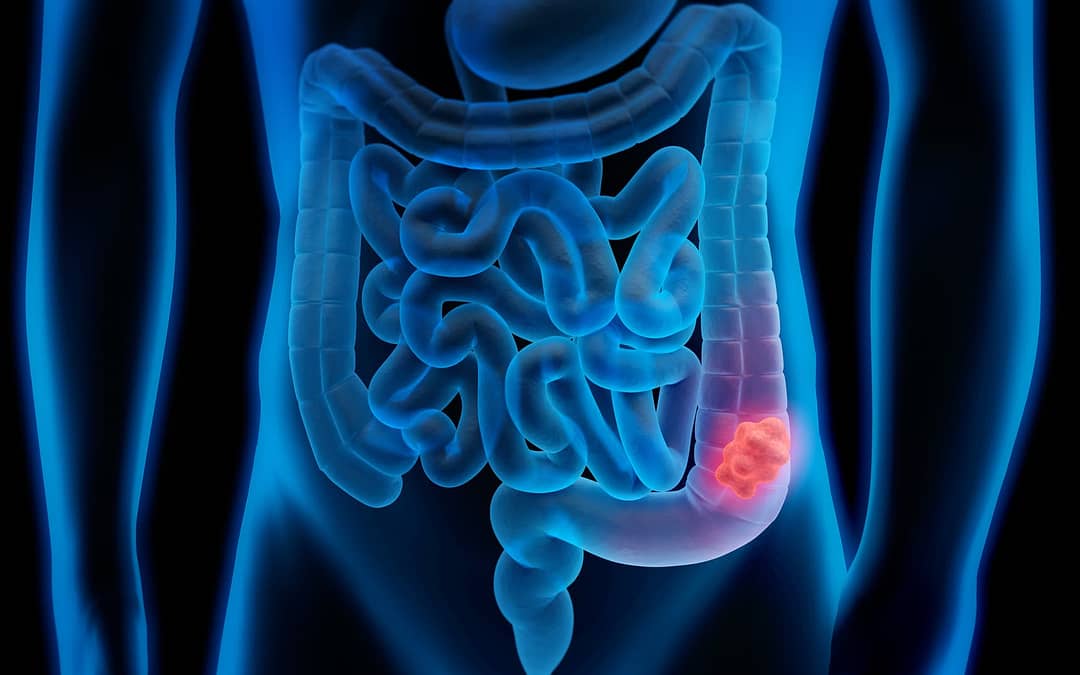
During a visit with a new primary care physician, a 45-year-old man told his doctor that he had a family history of cancer and Lynch syndrome, a hereditary condition that increases the risk for colon, uterine, and other cancers. He also recounted that he had tested negative for Lynch syndrome in 2013, and as a result, he had stopped having colonoscopy screenings since it seemed he didn’t have a familial condition and he hadn’t reached the age for colon cancer screening for the general population.
The physician referred him to a genetic counselor to ensure he would receive the appropriate follow-up care. The counselor reviewed the patient’s medical and family history, including prior genetic testing records. The patient’s records showed a family history of various cancers. The patient’s maternal grandmother was diagnosed at age 50 with breast cancer; his paternal grandfather had prostate cancer in his 60s; and his sister, father, and paternal aunt were reported to have had Lynch syndrome.
To read the full article, click here.